Case Study: Novo Nordisk and the Global Success of Ozempic
26.08.2025Authors: Miloš Stevanović, Božana Cvijić, Slaviša Lakić and Duško Karišik
Ozempic (semaglutide) comes in the form of a pre-filled injection pen; it was originally developed for type 2 diabetes but became globally popular as a weight-loss therapy.
The Danish pharmaceutical company Novo Nordisk has gone through an extraordinary journey from an insulin manufacturer to a global giant thanks to revolutionary drugs for diabetes and obesity. Their most famous product, Ozempic (active substance semaglutide), quickly gained worldwide fame and led to the “Ozempic mania” in the pharmaceutical market. Ozempic was initially developed for the treatment of type 2 diabetes, but it was shown that it significantly helps with weight loss, which caused a global craze for this drug. As a result, Novo Nordisk experienced accelerated revenue growth and market value, becoming the most valuable European company. In this report, we analyze the chronology of Novo Nordisk’s rise with financial indicators, the role of Ozempic in the diabetes and weight-loss market, global demand and trends related to this drug, competitors’ attempts to replicate its success, the situation in the market of Bosnia and Herzegovina, and a comparison of Ozempic with alternative therapies – with advantages and challenges listed.
Chronology of Novo Nordisk’s Growth
Novo Nordisk was founded back in 1923 in Copenhagen, through the merger of two Danish laboratories specialized in insulin production. For decades, the company built its reputation as a global leader in diabetes treatment, holding about half of the world’s insulin production. A key turning point occurred in the late 2010s when Novo Nordisk developed semaglutide, an innovative analog of the GLP-1 hormone that regulates blood sugar levels and appetite. Semaglutide was approved in 2017 in injectable form under the name Ozempic for type 2 diabetes therapy. At first, it passed relatively unnoticed in the wider circle because it was intended primarily for diabetics, but soon it was noticed that patients on Ozempic significantly lost weight. This led to the development of a stronger dose of semaglutide for obesity under the name Wegovy (approved in 2021), with which Novo Nordisk entered the vast obesity treatment market.
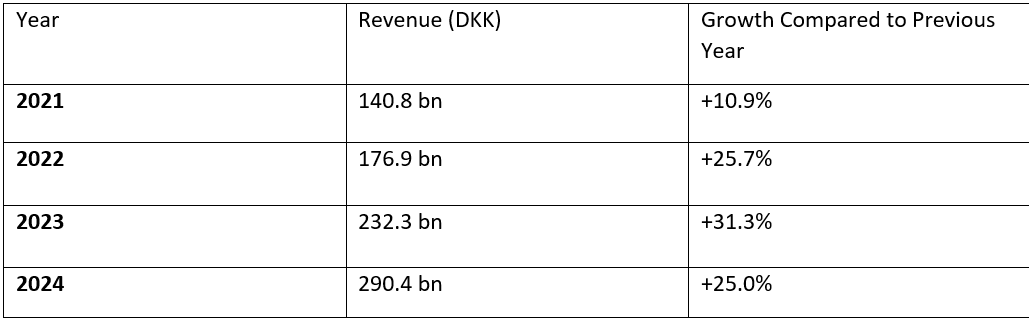
Since the introduction of semaglutide, the company’s financial indicators have exploded. Novo Nordisk’s revenues increased from about 141 billion DKK (Danish kroner) in 2021 to 232.3 billion DKK in 2023, with annual growth rates of 25–31%. In 2024, revenue reached 290.4 billion DKK (Table 1), which is more than double compared to the period before Ozempic. At the same time, the company’s net profit has been rapidly growing (a jump of 45% in the first half of 2023 on an annual basis), driven by demand for semaglutide-based drugs. Ozempic and Wegovy together accounted for about 55% of Novo Nordisk’s total revenues in 2023, illustrating how much impact this single innovative product had on the company’s business.
Table 1: Novo Nordisk revenue growth 2021–2024 (in billions of DKK)
The huge popularity of Ozempic and Wegovy directly affected the stock value of Novo Nordisk. From 2018 to 2023, the company’s market capitalization quadrupled, reaching $419 billion in 2023. By that time, Novo Nordisk overtook LVMH and became the most valuable European company on the stock exchange.
This success was driven by global demand for semaglutide, which analysts estimate could bring $100 billion in annual revenues worldwide by 2030. Novo Nordisk itself is rapidly increasing production capacities, but even with the expansion of factories in Denmark, the United States, and France, it still cannot keep up with demand. This situation additionally increases investor optimism, raising the price of shares and strengthening Novo Nordisk’s dominant position in the market.
The Role of Ozempic in the Diabetes and Weight-Loss Market
Ozempic was initially positioned as a modern therapy for type 2 diabetes, especially for patients who did not achieve sufficient regulation of blood sugar with metformin or insulin. Its mechanism of action is based on the stimulation of GLP-1 receptors, which increases insulin secretion, reduces glucagon secretion, and slows gastric emptying – leading to better glycemic control.
However, clinical studies and experiences in practice showed that patients also lose significant amounts of body weight – up to 15% of body mass. This opened up new possibilities for using semaglutide in the treatment of obesity, a condition that affects more than 650 million adults worldwide. Wegovy, as a version of semaglutide intended exclusively for weight reduction, became a “game changer” in medicine and the pharmaceutical industry.
The obesity treatment market is estimated at $100 billion annually, and experts predict that GLP-1 receptor agonists like Ozempic and Wegovy will dominate this area in the coming decade. Novo Nordisk thus secured leadership both in diabetes therapy and in the fight against obesity, two diseases that represent key challenges for global public health.
Global Demand and Trends
The demand for Ozempic and Wegovy exceeded all expectations. In many markets, patients are on waiting lists, while doctors face pressure from patients who request prescriptions even when they are not medically indicated. Social networks, especially TikTok and Instagram, played a big role in popularizing Ozempic as a “miracle drug for weight loss,” which led to increased demand among celebrities, influencers, and the general public.
Novo Nordisk faced difficulties in securing sufficient production capacity. Despite the construction of new factories and multimillion-dollar investments in expanding facilities, supply remains limited compared to demand. Because of this, in certain markets, including the United States and the European Union, authorities restricted the use of Wegovy and Ozempic exclusively to patients with medical indications, primarily diabetics and those with serious obesity problems.
Global trends also show that the success of semaglutide has triggered a “gold rush” in the pharmaceutical industry. Competitors such as Eli Lilly are developing their own drugs based on similar mechanisms of action. Eli Lilly launched tirzepatide under the brand names Mounjaro (for diabetes) and Zepbound (for weight loss), which showed even better effects in some studies. Analysts expect intense competition between Novo Nordisk and Eli Lilly in the coming decade, as both companies race to dominate the market for obesity therapy.
Competitors’ Attempts and Market Challenges
The enormous commercial success of Novo Nordisk’s products has prompted global competitors to enter the race. Eli Lilly, as the most serious rival, introduced tirzepatide, a drug that acts as a dual agonist of GLP-1 and GIP receptors. Under the names Mounjaro (for diabetes) and Zepbound (for obesity), this drug showed in clinical trials even greater effectiveness in reducing body weight compared to semaglutide. Analysts predict that the rivalry between Novo Nordisk and Eli Lilly could dominate the pharmaceutical industry in the next decade, as both companies invest billions in research, development, and production capacities.
In addition to Eli Lilly, other pharmaceutical companies such as Pfizer, Amgen, and AstraZeneca are trying to develop their own therapies for obesity and diabetes, but they are still in earlier phases of clinical trials. This indicates that the GLP-1 segment could become one of the most competitive areas in medicine.
Nevertheless, Novo Nordisk currently maintains a significant advantage thanks to the strong position of Ozempic and Wegovy, the already established production and distribution system, and the huge patient base. The main challenges facing the company remain the limitations in production capacity, potential side effects of the drugs (such as nausea, vomiting, and increased risk of pancreatitis), and ethical debates about the non-medical use of these drugs for cosmetic purposes.
Situation in Bosnia and Herzegovina
Ozempic in BiH was registered relatively quickly after the world premiere – according to the data of the Agency for Medicinal Products and Medical Devices of BiH (ALMBiH), the authorization for placing it on the market in BiH was obtained in August 2019. The medicine is available exclusively on a doctor’s prescription and was initially indicated for the therapy of type 2 diabetes. In Bosnia and Herzegovina, Wegovy (semaglutide 2.4 mg for obesity) is currently not registered, which means that semaglutide is also used for weight loss off-label through Ozempic. The oral form Rybelsus (semaglutide tablets) is also registered, but injectable Ozempic is still far more sought after.
A big step towards availability happened at the beginning of 2025 – Canton Sarajevo included Ozempic on its List A of essential medicines, which means that it is fully covered by health insurance funds in this canton. From the adoption of the new cantonal list of medicines, patients with type 2 diabetes in CS can receive Ozempic free of charge, under certain conditions. According to the criteria of the Health Insurance Institute of CS, therapy may only be initiated by an endocrinologist or internist-diabetologist in a tertiary institution, and only in patients in whom other options have been exhausted. In addition, the patient must also continue to follow dietary and physical activity measures, and before switching to Ozempic, the effect of the previous therapy is monitored for six months. Strict criteria are also prescribed for the continuation of therapy: after 6 months on Ozempic, success is evaluated – a reduction in HbA1c of at least 0.5% and a loss of ≥5% of body weight is required for insurance to continue covering therapy. Such criteria ensure that the drug is given to those who truly benefit in controlling diabetes.
The retail price of Ozempic in BiH is relatively high. According to the information of the Institute, the reference price on the list amounts to 148.81 KM + VAT per package. When patients purchase the drug independently (outside of insurance), the final price may vary by pharmacy due to margins and other factors. Comparatively, this is approximately €80 per package, which is significantly more favorable than in some Western markets (in the USA the full price of Ozempic is allegedly around USD 1000 per month, which has also provoked political criticism of high prices). However, even 150–200 KM per month represents a significant expense for the average patient in BiH, so the inclusion on the positive list in CS is an important step forward. It is still not clear when other cantons in the Federation of BiH, as well as Republika Srpska, will introduce semaglutide on their lists of medicines, but given the clinical effectiveness, broader availability can be expected in the future.
Since the appearance of Ozempic on the BiH market in 2023, a sudden expansion of interest among patients has been recorded, including those who do not have diabetes but want to lose weight. Media reports state that citizens began to “massively” request this drug from doctors because of excess weight, under the influence of stories about the miraculous effect from the USA and social networks. This trend worried experts: endocrinologists warned that unnecessary use of Ozempic carries risks, and at the same time endangers the supply for diabetics who need it. Indeed, shortages of Ozempic began to occasionally appear in BiH pharmacies during 2023, which is linked to the growth in demand. Although there are no publicly available precise data on imported quantities, it is indicative that doctors and patients reported occasional procurement problems.
ALMBiH reacted to this trend by issuing a public statement in November 2023, warning citizens that Ozempic may be used exclusively under medical supervision and only in approved indications (therefore for type 2 diabetes), and that any “self-directed” use for weight loss can be dangerous. The Agency especially emphasized that the medicine is procured exclusively legally, in pharmacies, since cases were recorded in the region (Austria) where patients ended up in hospital due to using counterfeit Ozempic purchased via the internet. In the statement it is reminded that Ozempic is a medicine subject to strict control (prescription-only regime) and that any attempt to purchase outside the authorized supply chain (wholesaler-pharmacy) increases the risk of adverse consequences. ALMBiH called on patients to be cautious, and pharmacists and doctors to respect regulations, in order to prevent misuse of this drug.
The regulations in BiH follow European standards also regarding the safety of semaglutide use. In June 2025 ALMBiH transmitted a new warning from the World Health Organization (WHO) about a potential rare side effect of semaglutide – non-arteritic ischemic optic neuropathy (NAION), which can cause sudden loss of vision. Although this risk is extremely low, healthcare professionals were ordered to carefully monitor patients on Ozempic and report suspicions of this side effect. This example shows that pharmacovigilance is actively implemented in BiH and that the competent institutions are engaged in monitoring the safety of new medicines.
Summarized, Ozempic has been present in BiH for several years and is used mainly in diabetes, with growing interest also for off-label use in obesity. Availability is gradually improving (first through cantonal lists), but at the same time precautionary measures are being implemented to prevent misuse and ensure that the medicine is used by those who truly need it.
Comparison of Ozempic with Alternative Medicines: Advantages and Challenges
Ozempic (semaglutide) today faces competition within the wider class of GLP-1 agonists and new therapies for obesity. Below are highlighted the main similarities and differences, advantages and challenges in comparison with some key alternatives:
Wegovy (semaglutide 2.4 mg) – This is the same active ingredient as Ozempic, but in a higher dose and approved exclusively for the treatment of obesity. The advantage of Wegovy is that it achieves an even greater average weight loss (about 15% of body weight in clinical studies) and is intended for persons with a BMI ≥30 or ≥27 with comorbidities. This provides targeted therapy for patients with severe obesity. Challenges of Wegovy include limited availability (frequent shortages due to huge demand) and high price. Also, Wegovy is only gradually being introduced to markets outside the USA – in BiH it is not yet available, so the effects of Wegovy here manifest indirectly through pressure on Ozempic.
Mounjaro/Zepbound (tirzepatide) – A medicine by Eli Lilly that combines GLP-1 and GIP agonistic action. Studies have shown impressive results in blood sugar control and weight reduction (even over 20% in some patients), which makes tirzepatide potentially more effective than semaglutide. The advantage of tirzepatide is precisely this double target – it acts on two appetite hormones which can give a stronger effect. Lilly has already launched Mounjaro for diabetes, and approval (under the name Zepbound) for obesity is expected soon, which will give it broader application. The challenge for Ozempic is that tirzepatide will probably take part of the market, especially among patients who seek maximum weight loss. Nevertheless, tirzepatide shares similar side effects (nausea, vomiting, digestive issues) and so far does not have long-term experience in widespread use. From the patient’s perspective, the appearance of tirzepatide provides a new option – those who do not respond well to Ozempic may have a better response to the dual therapy. For healthcare systems, competition could also mean more favorable prices in the future.
Other GLP-1 agonists (liraglutide, dulaglutide, etc.) – Before Ozempic, medicines such as Victoza (liraglutide, daily injection) and Trulicity (dulaglutide, weekly injection) were in use for type 2 diabetes. The advantages of these older medicines are that they have been well studied over a longer period of years and have established safety profiles. Some (such as liraglutide under the name Saxenda) were also approved for weight loss, but require daily injections and the weight loss is more modest. Compared to them, Ozempic has a clear advantage: it is given only once a week and causes a greater drop in glycated hemoglobin and greater weight reduction. The challenge for older GLP-1 analogues is that semaglutide and its successors will practically push them out of use except in cases where the newer ones are not available or are contraindicated. The only trump card of older medicines could become price – as their patents expire, cheaper generic versions may appear earlier than for semaglutide.
Oral medicines and new therapies – Rybelsus (oral semaglutide) already exists, which offers the advantage of taking a pill instead of an injection. However, Rybelsus must be taken every day on an empty stomach and its absorption is variable, so in practice it is somewhat less effective than the injectable form. The advantage is the comfort for the patient who does not want injections, but the challenge remains to achieve equal clinical outcomes. Completely new agents are also in development – e.g. glucagon receptor agonists or triple agonists (acting simultaneously on GLP-1, GIP, and glucagon) which could provide an even stronger effect on weight loss. Less invasive forms are also being explored (next-generation pills, transdermal patches, etc.). As new therapies appear, Ozempic will face the challenge of maintaining its status as the “gold standard.” Its advantage is the strong evidence base and the experience of millions of patients, but the risks include the possibility of new side effects (such as the recently discovered very rare NAION – vision damage) and the competition of innovations that could surpass existing results.
Safety profile and tolerance – Ozempic and similar GLP-1 medicines share mostly similar side effects. The most common are gastrointestinal problems: nausea, vomiting, diarrhea, or constipation, especially in the first weeks of therapy while the body adapts. These side effects are usually mild to moderate and transient. More serious possible side effects being monitored include pancreatitis (inflammation of the pancreas) and the risk of gastric paralysis due to delayed stomach emptying. Recently, as mentioned, the association with a rare eye complication (NAION) is also being considered. All these medicines are not recommended for pregnant women nor for people with some specific gastrointestinal diseases. In the context of comparison, no dramatic difference has been observed in the side effect profile between Ozempic and tirzepatide – both belong to the most effective weight loss medicines to date, but at the cost of moderate tolerance (many users experience at least some form of digestive discomfort). For patients it is crucial that these therapies are taken under professional supervision, with gradual dose titration, in order to minimize side effects.
In short, Ozempic has set a new standard in the treatment of diabetes and obesity, but competition is heating up on multiple fronts. Its advantages are proven high efficacy, simple weekly use, and a wide spectrum of benefits (lowering blood sugar, weight, and potentially organ protection). Challenges come in the form of: (1) new, even more potent medicines (such as tirzepatide) that may reduce its market share, (2) high therapy costs and public/regulatory pressure to lower prices, (3) logistical limitations in production and distribution given the enormous demand, and (4) the need to monitor long-term safety in the wider population of users.
Conclusion
Novo Nordisk and its blockbuster drug Ozempic represent a paradigmatic example of how a single innovation can transform both a company and the entire medicines market. In the span of only a few years, semaglutide grew from just another new diabetes medicine into a globally sought-after preparation that drives a multibillion-dollar market and changes the approach to treating obesity. The company Novo Nordisk achieved astronomical growth of revenue and value, at the same time becoming an important factor also in the wider economic context (contribution to Denmark’s GDP, employment, taxes). Ozempic and Wegovy brought medical benefits not only to diabetics but also to people with serious overweight, with promising indications of improvements in other health parameters.
However, the incredible demand for semaglutide also opened some questions: how to ensure fair availability of the medicine, prevent misuse and counterfeits, and how to educate the public that this is not a miracle drink but a serious medicine with possible side effects. The appearance of the “Ozempic mania” also demonstrated the power of social networks in shaping the demand for medicines, which posed a new task for regulatory bodies.
Competing pharmaceutical giants are rapidly developing their own alternatives – which is positive from the standpoint of progress in medicine and therapy availability, but represents a challenge for Novo Nordisk’s dominance. In Bosnia and Herzegovina, the experience with Ozempic reflects global developments: the drug was greeted with enthusiasm, but with the caution of regulators to ensure it is applied correctly and without risks for patients.
The general lesson of this case is that innovation in pharmacy can have far-reaching effects – improve population health, boost the economy, but also trigger social phenomena that must be carefully managed. Novo Nordisk, with its tradition in diabetes and bold step into the field of obesity, demonstrated how strategic investments in research can lead to a medicine that changes the rules of the game. The continuation of the story will depend on the company’s ability to stay ahead of the competition, continue innovating, and at the same time ensure that the benefits of its medicines are safely and ethically available to all who need them.
/ / /
"Standard Prva" LLC Bijeljina is a company registered in Bijeljina at the District Commercial Court in Bijeljina. Company’s activities are accountancy, repurchases of receivables, angel investing and other related services. Distressed debt is a part of the Group within which the company repurchases the receivables, which function and are not returned regularly.
Lawyer’s Office Stevanović is the leading lawyer’s office in the region with the seat in Bijeljina. The LO abbreviation represents Lawyer’s Office of Vesna Stevanović and Lawyer’s Office of Miloš Stevanović.
Contact for media press@advokati-stevanovic.com or via telephone 00 387 55 230 000 or 00387 55 22 4444.




